
Ca has a higher IE 1 because its radius is smaller. The third ionization energy for the ion formed was found to be 2,700 kJ/mol. The second ionization energy for the ion formed was found to be 1,800 kJ/mol. Explain, Hint: Look at the valence configurations, and recall that half-filled and fully filled subshells have extra stability. Using experimental data, the first ionization energy for an element was found to be 600 kJ/mol. a) The first ionization energy of sodium is greater than rubidiums because sodium is on the third. c) The second ionization energy for K is greater than the second ionization energy of Ca. b) The atomic radius of Li is larger than that of Be. Match the elements in the left column to the appropriate blanks in the sentences on the right. Free Response: a) The first ionization energy of Na is greater than the first ionization energy of Rb. Reset Help S Of Ge or Br, the element with the higher. 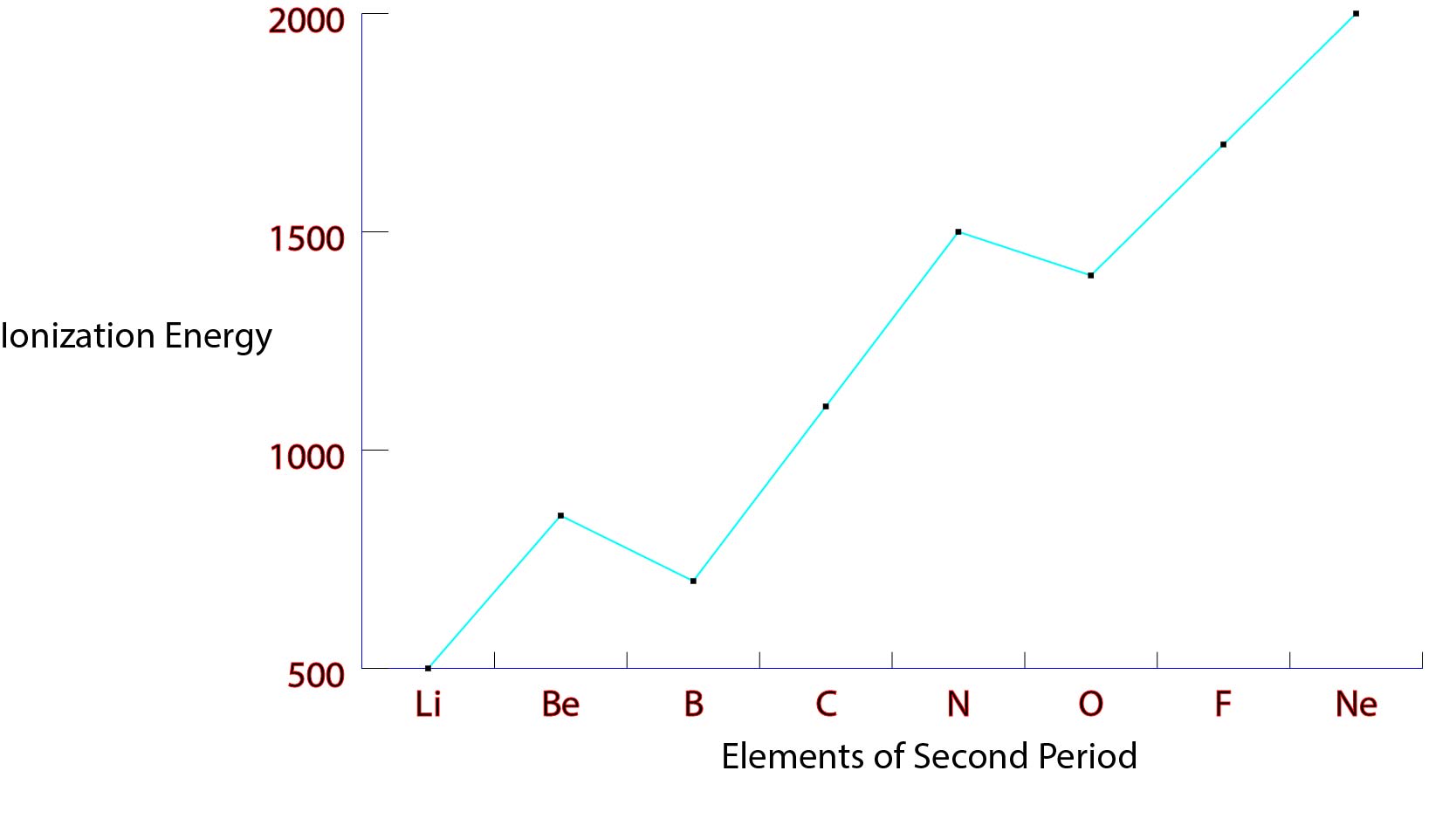
Make certain each sentence is complete before submitting your answer. 
Match the elements in the left column to the appropriate blanks in the sentences on the right. But in period 2, Be has a higher first ionization energy than B, and N has a higher first ionization energy than O. Transcribed Image Text: Part A Choose the element with the highest first ionization energy from each of the following pairs. On the basis of periodic trends, determine the element with the higher first ionization energy in each pair (if possible). But in period 2, Be has a higher first ionization energy than B, and N has a higher first ionization energy than O. First ionization energies tend to increase across a period. After 1 valence electron of K is lost, the whole fourth energy level is lost as well, meaning that now K acts as Argon and experience less electron shielding than Ca does, making the second ionization energy of K much higher than Ca. First ionization energies tend to increase across a period. Based on position in the periodic table, which element of the following pairs has the higher first ionization energy O, Ne Ca, Sr K, Cr Br, Sb In, Sn 5. Therefore, Be has a higher effective nuclear charge and is able to pull the c) The second ionization energy of K is greater than Ca because even though they are both on the fourth energy level and experience the same amount of electron shielding, k only has 1 valence electron while Ca has 2. b) The atomic radius of lithium is larger than Be because even though tey are both on the second energy level and experience the same amount of electron shielding, Be has 4 protons whereas Li only has three protons. Although rubidium has 37 protons while sodium only has 11 protons, the lections shielding in rubidium I greater than sodium, requiring more energy for a valence electron to be removed from sodium. A) The first ionization energy of sodium is greater than rubidium's because sodium is on the third energy level and does not experience as much electron shielding as rubidium does on the fifth energy level.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
